EXCEPTIONAL
RESULTS

Dr. Larisse Lee
Dr. Larisse Lee is a board certified vascular surgeon who trained at Stanford University, Massachusetts General Hospital, a leading Harvard hospital, and Cornell-Columbia Presbyterian Hospitals in New York City.
Dr. Lee established the L.A. Vein Center to provide the best comprehensive venous care to the greater Los Angeles area. Dr. Lee received the 2020 “Ultimate Vein Treatment” Award.



Our Services
Why Choose Us
Board-certified in both Vascular Surgery and General Surgery, Dr. Lee has received numerous honors and awards, including a National Research Service Award bestowed by the National Institutes of Health. Dr. Lee has conducted several studies in vascular surgery to increase knowledge and understanding in the field.
• Latest techniques in vein removal
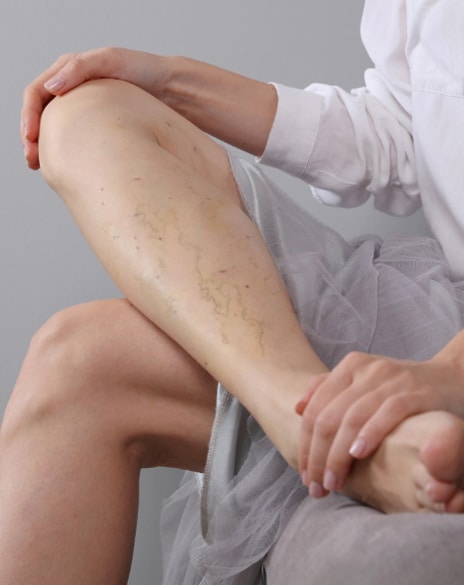
• Varicose vein and spider vein treatment
• Xeomin, Radiesse, Belotero
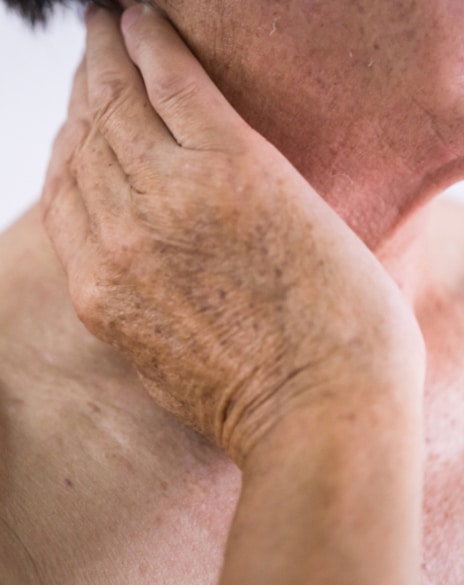
• Minimally invasive cosmetic and medical procedures
• In house ultrasound evaluation
• Focus on patient safety, education and results
• Harvard Stanford trained board certified surgeon







Media & TV Appearances
Dr. Larisse Lee is one of the most widely regarded vein specialists in Los Angeles.



What Our Patients Are Saying
View Our Gallery
Browse Through Our Gallery Of Patient Results